XCELCAREER is the best institute to do the Clinical Research certification course. We are the top-rated institute on Google.
Get to Know More: https://xcelcareer.com/


CLINICAL DATA MANAGEMENT COURSE
What is Clinical Data Management?
When new drugs or devices are tested in humans, the data generated by and related to these trials is known as clinical data.
Read More: https://xcelcareer.com/clinica....l-data-management-co
Definition of Clinical Data Management:
Clinical data management (CDM) is a critical phase in clinical research, which leads to generation of high-quality, reliable, and statistically sound data from clinical trials. Clinical data management assures collection, integration and availability of data at appropriate quality and cost.
CLINICAL DATA MANAGEMENT is a process of ensuring that data collected during a clinical trial is…
Accurate
Complete
Logical
Consistent
Objectives of CLINICAL DATA MANAGEMENT:
To Ensure:
That collected data is complete & accurate so that results are correct.
That trial database is complete, accurate & a true representation of what took place in trial.
That trial database is sufficiently clean to support statistical analysis & its subsequent presentation & interpretation.
Clinical Data Management Overview
Clinical Trials and drug discovery introduction
Principles of Clinical trials
21CFR part 11 introduction
GCP (Good Clinical Practice Principles
Clinical Data Management Introduction
Clinical Data Management phases
Start-up Phase introduction. Conduct Phase introduction
Discrepancy Management
Data set validation review
Offline Listing review
All third-party vendor reconciliation
SAE reconciliation
Study close-out phase introduction and activities

CLINICAL DATA MANAGEMENT COURSE
What is Clinical Data Management?
When new drugs or devices are tested in humans, the data generated by and related to these trials is known as clinical data.
Read More: https://xcelcareer.com/clinica....l-data-management-co
Definition of Clinical Data Management:
Clinical data management (CDM) is a critical phase in clinical research, which leads to generation of high-quality, reliable, and statistically sound data from clinical trials. Clinical data management assures collection, integration and availability of data at appropriate quality and cost.
CLINICAL DATA MANAGEMENT is a process of ensuring that data collected during a clinical trial is…
Accurate
Complete
Logical
Consistent
Objectives of CLINICAL DATA MANAGEMENT:
To Ensure:
That collected data is complete & accurate so that results are correct.
That trial database is complete, accurate & a true representation of what took place in trial.
That trial database is sufficiently clean to support statistical analysis & its subsequent presentation & interpretation.
Clinical Data Management Overview
Clinical Trials and drug discovery introduction
Principles of Clinical trials
21CFR part 11 introduction
GCP (Good Clinical Practice Principles
Clinical Data Management Introduction
Clinical Data Management phases
Start-up Phase introduction. Conduct Phase introduction
Discrepancy Management
Data set validation review
Offline Listing review
All third-party vendor reconciliation
SAE reconciliation
Study close-out phase introduction and activities

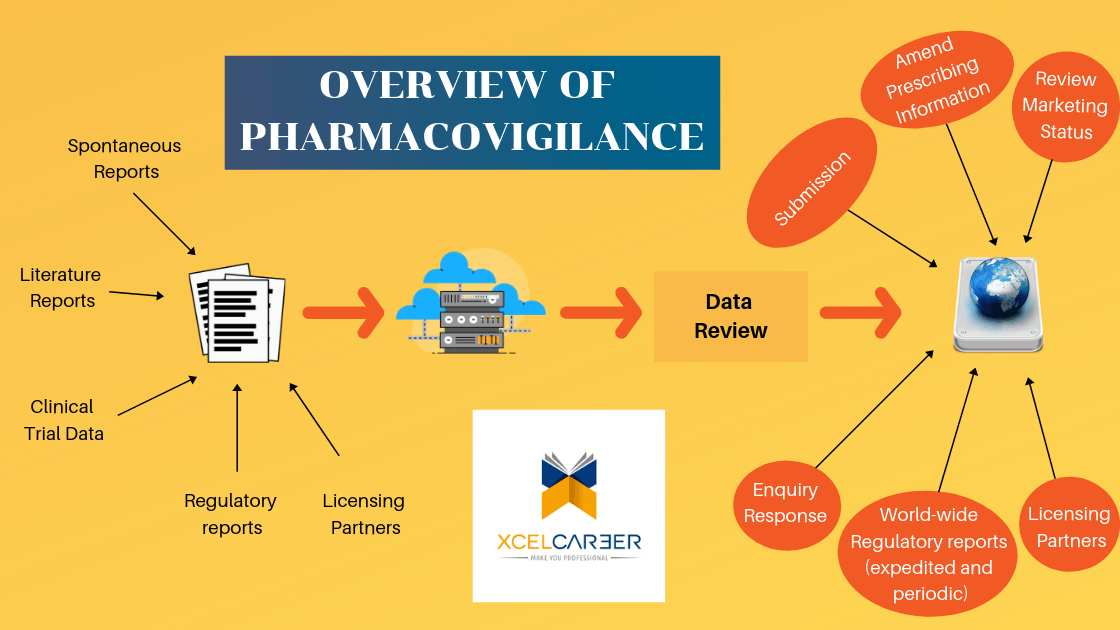
Overview of the Pharmacovigilance Course
https://xcelcareer.com/pharmacovigilance-course/
Drug safety or Pharmacovigilance is in the spotlight of new drug development. This carves a vital niche inside the expanding pharmaceutical business. It would be apt to say that as much as the pharmaceutical business is evergreen, pharmacovigilance is ever greener.
In the USA the total cost incurred due to medication errors or adverse drug reactions (ADRs) exceeds 136 billion USD annually and hence, the regulatory agencies worldwide have developed stringent guidelines for drug approvals as well as for continuous monitoring of drugs in clinical practice.
The World Health Organization (WHO) has defined pharmacovigilance as the science and all the associated activities related to the detection, evaluation, understanding and prevention of ADRs or any other drug-related problems. ADRs (especially the serious drug reactions or SAEs), medication and prescription errors and poor drug qualities are the major concerns which are dealt with by pharmacovigilance systems. Thus, risk management, risk mitigation plans and discovery of safety signals from data mining of existing safety databases are immensely important activities during the development as well as post marketing phases in the life cycle of a drug. Pharmacovigilance is an important and integral part of clinical research and these days it is growing in many countries and many pharmacovigilance centers are working for drug safety monitoring in this global pitch.
A very important constituent of a compliant pharmacovigilance program is to maintain large safety databases. Such requirements of maintaining and analyzing extremely large quantities of data has pushed pharmaceutical companies to outsource pharmacovigilance data management and analysis to cost effective hubs like India, Philippines, China, to name a few major ones. It is pertinent to note that the outsourced pharmacovigilance activity to India is on a steep rise for last 5 years. With the heavy outsourcing comes the need of trained professionals, and so XCEL CAREER
Objective of Pharmacovigilance
Proactive monitoring and reporting on the quality, safety and efficacy of drugs,
Assessment of the risks and benefits of marketed medicines,
Monitoring the impact of any corrective actions taken,
Providing information to consumers, practitioners and regulators on the effective use of drugs,
Designing programs and procedures for collecting and analysing reports from health care professionals (HCPs), patients, relatives, lawyers, journalists etc.
Early detection of unknown/unexpected safety problems,
Detection of increases in frequency of Adverse Drug Reactions (ADRs) to a drug, in patients/subjects treated with this medication,
Identification of risk factors for ADRs,
Risks Analysis Mitigation,
Benefit-Risk balance.

pro yash
Eliminar comentario
¿ Seguro que deseas eliminar esté comentario ?